Toward the Two-Step Microdynamic Phase Transition Mechanism of Oligo(Ethylene Glycol) Methacrylate-Based Copolymer...
Citation
Yalan Dai, and Peiyi Wu*. Toward the Two-Step Microdynamic Phase Transition Mechanism of Oligo(Ethylene Glycol) Methacrylate-Based Copolymer with a LCST-Type Poly(Ionic Liquid) Block. Phys. Chem. Chem. Phys. 2017, 19, 18556-18564.
Abstract
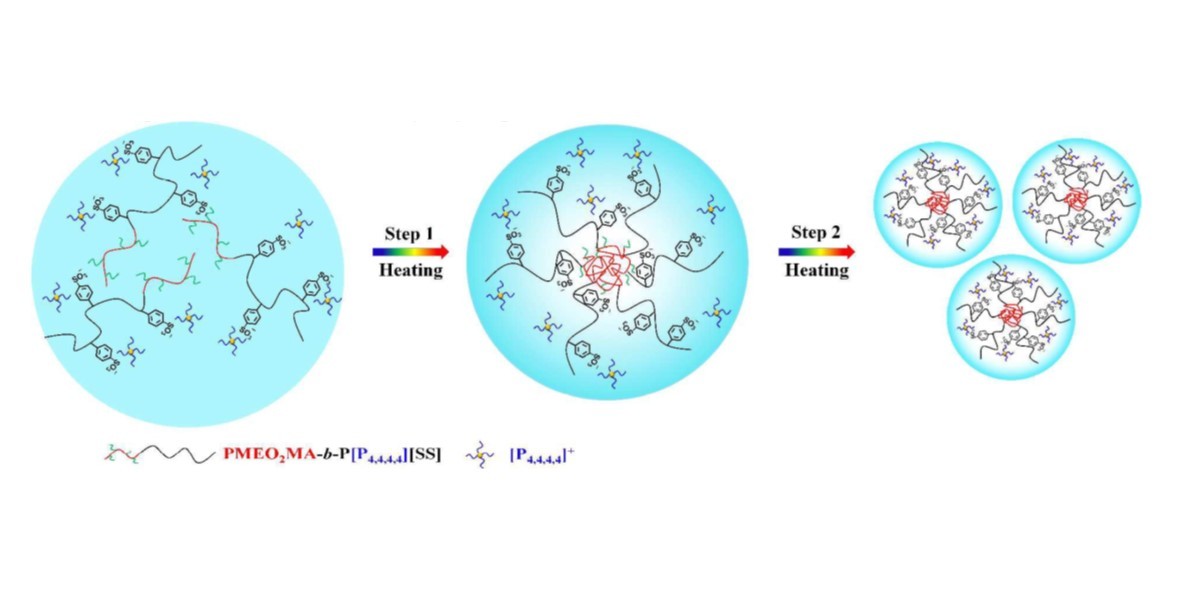
A new LCST-type thermoresponsive polyelectrolyte P[P4,4,4,4][SS], poly(tetrabutyl phosphonium styrene sulfonate), was introduced to PMEO2MA (poly(2-(2-methoxyethoxy)ethyl methacrylate)) by RAFT polymerization, to explore the transition behavior of the block copolymer PMEO2MA-b-P[P4,4,4,4][SS] with two distinct LCST-type segments. A relatively sharp LCST-type phase transition with only one transition point is observed in turbidity curves, while the whole phase transition is completely different from the micro perspective. The phase transition temperature range is relatively broad with the unsynchronized changes of different protons of the two blocks in the temperature-variable 1H NMR analysis. From PCMW analysis, it is found that there exists an obvious two-step phase transition behavior, especially in the region of C-H groups. Accordingly, we divided the whole transition process into two subregions: 20-40 oC and 40-55 oC in 2Dcos analysis. At the first stage of 20-40 oC, the CH3 groups mainly belonging to the backbones of PMEO2MA blocks have an earliest response towards the heating and drive the first step of the dehydration process of PMEO2MA-b-P[P4,4,4,4][SS], resulting in the formation of an intermediate micelle state comprised of the collapsed PMEO2MA core and hydrophilic P[P4,4,4,4][SS] corona. In particular, due to the conformational changes and the more compact structures, the interaction between C=O groups and P[P4,4,4,4][SS] segments (v(C=O…D2O-PILs)) was observed by IR analysis. With the rising of the temperature continually, when it reaches in the second temperature range of 40-55 oC, the P[P4,4,4,4][SS] segments start to collapse and expel the water molecules out, driven by the anions of the poly(ionic liquid)s, with the phosphonium cations distributed in the relatively hydrophilic outside.